Case Presentation:
A 66-year-old man with a history of HTN, HLD, and obesity presents to the emergency department with one week of exertional shortness of breath and intermittent pleuritic chest pain. He reports no history of similar symptoms. He has no leg swelling, orthopnea, or fevers. The patient recently sprained his ankle and notes that he has been significantly less active than usual.
Initial vitals in triage are remarkable for sinus tachycardia to115/bpm, oxygen saturation of 92% on room air, and a blood pressure of 145/72 mmHg. On physical exam, lungs are clear to auscultation bilaterally, no leg edema, and normal S1 and S2 heart sounds with regular rate and rhythm with no murmurs, rubs, or gallops; JVD is not appreciated.
Following these studies, laboratory studies are obtained and are notable for:
- BNP 369 pg/mL (normal <100)
- HS troponin 97 ng/L (normal <15-20)
- Cr 0.95 mg/dL
- Lactate 1.6 mmol/L
- Age-adjusted D-dimer is >20,000 micrograms/mL
Ask Yourself:
Questions:
What tools can be used to guide the appropriate use of diagnostic imaging when PE is on the differential diagnosis?
How are patients with PE risk-stratified, and how does this guide further treatment?
What is the role of catheter-based interventions for the treatment of PE?
How do we determine anticoagulation duration after PE ?
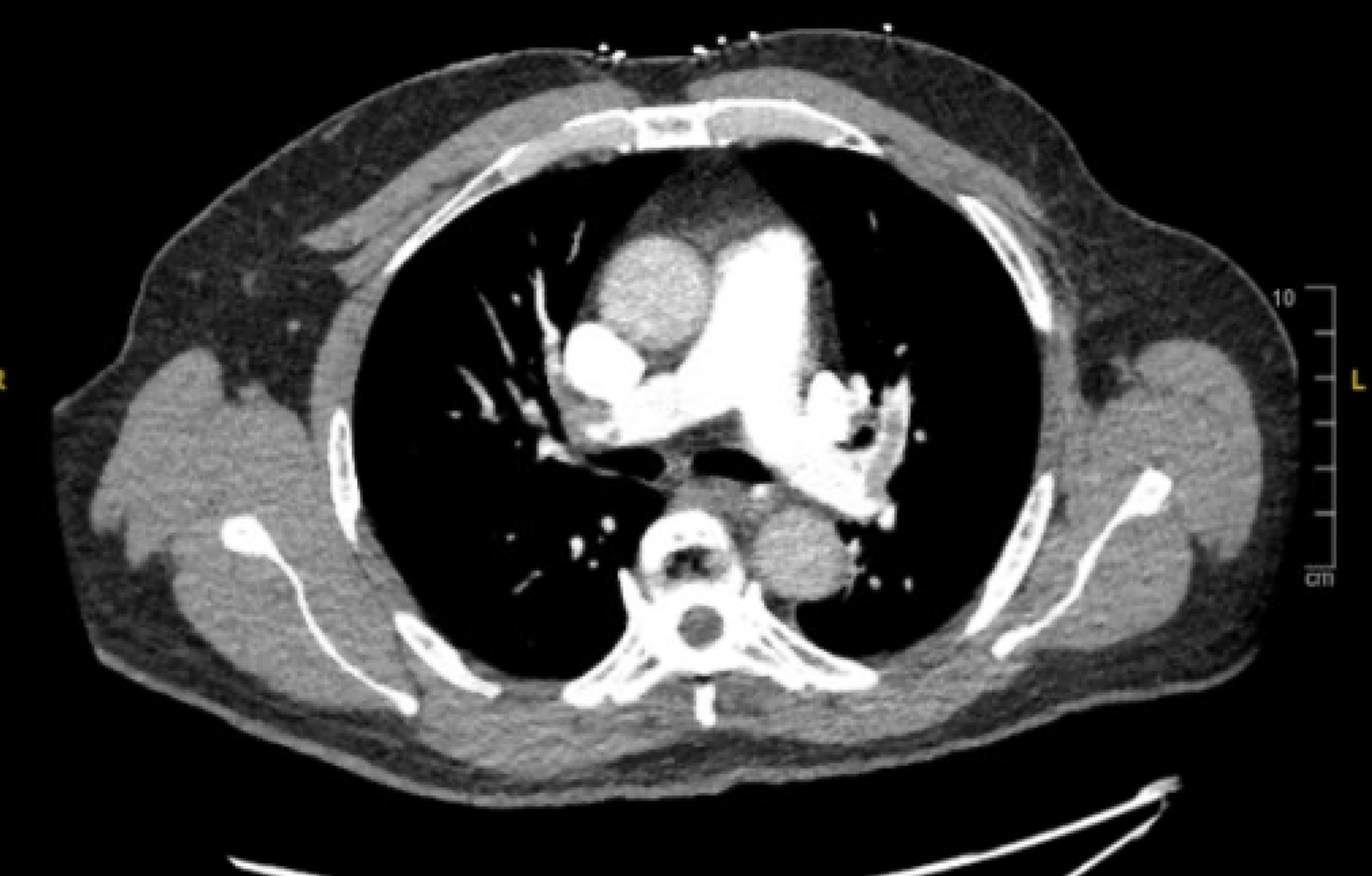
CT PE showing filling defects in the right and left pulmonary veins
Background
Pulmonary embolism (PE) occurs when a blood clot originating elsewhere in the body migrates to the pulmonary arterial circulation. PE accounts for 250,000 hospitalizations and 100,000 deaths per year in the United States. PE most commonly originates from lower extremity deep vein thrombosis (DVT). About 50% of lower extremity DVTs ultimately embolize to the lungs. Sometimes, there will be no residual lower extremity DVT appreciated in patients with PE as the entire thrombus may have mobilized to the lungs. Upper extremity DVTs are much less likely to result in PE, but embolization to the lung can still occur.
Common clinical risk factors to keep in mind for the development of PE include cancer, estrogen-containing contraceptives, obesity, cigarette smoking, recent air travel, surgery, trauma, and prolonged immobility.
Myocardial infarction, cerebral vascular accidents (CVA), and venous thromboembolisms (VTE) share some common risk factors, many of which are facilitated by the presence of inflammation, hypercoagulability, and endothelial injury. Due to this similar underlying pathophysiology, patients with VTE are more than twice as likely to have future myocardial infarction or CVA.
Clinical Presentation
The nonspecific symptoms of PE can make the diagnosis challenging, as many other pathologies can present similarly. These symptoms include dyspnea (the most common concern), fatigue, chest pain, fever, and hemoptysis. Pleuritic chest pain is a classic, but nonspecific, finding that may reflect pulmonary infarctions, typically from more distal pulmonary vascular occlusion. If an alternate diagnosis is initially suspected but there is a lack of response to adequate treatment, you should consider whether testing for PE is needed, as it can masquerade as other common conditions such as asthma or chronic obstructive pulmonary disease (COPD) exacerbation, pneumonia, pulmonary edema, or pericarditis. Additionally, while PE most often originates from the lower extremities, migration of the clot can result in a lack of overt lower extremity manifestations.
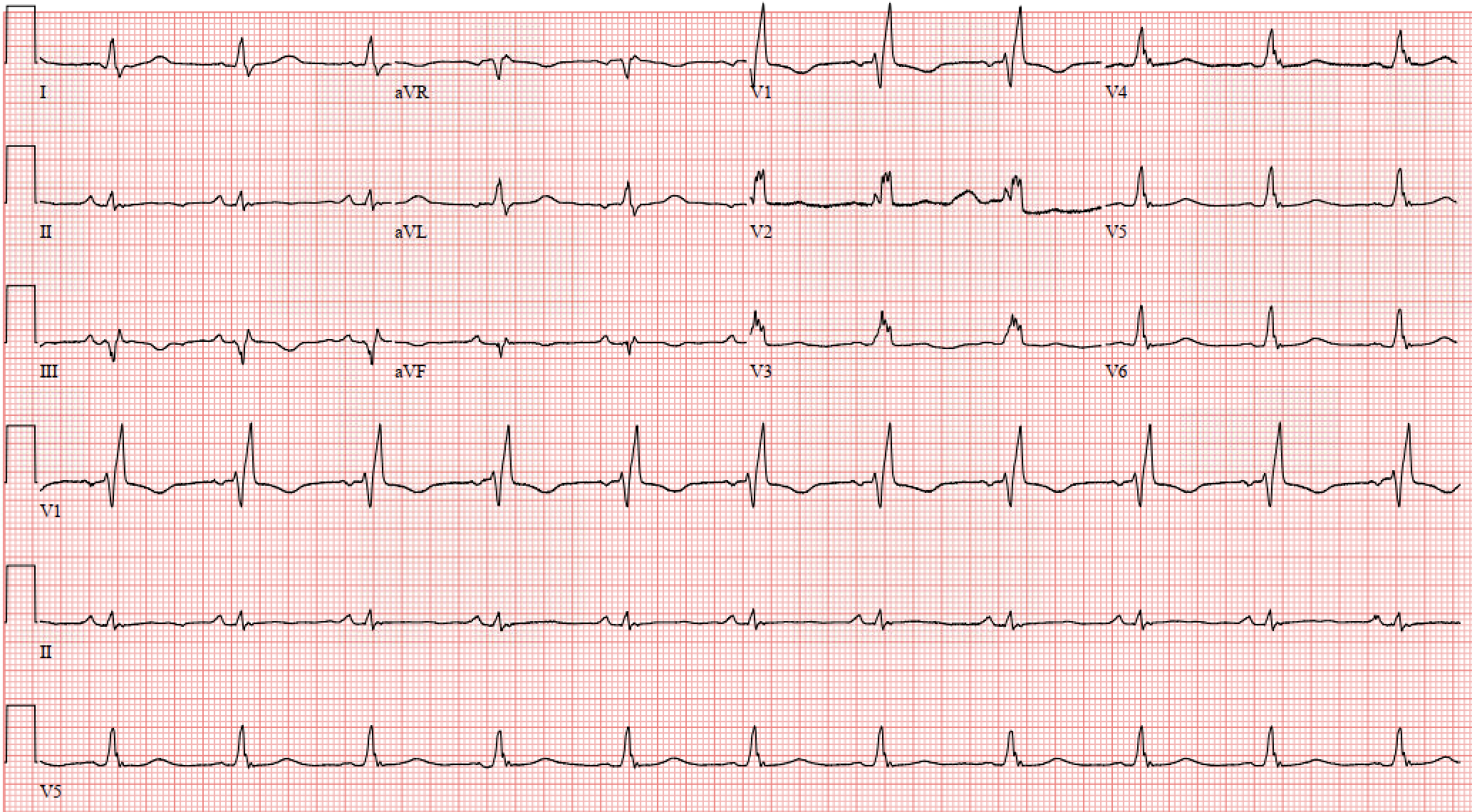
EKG showing new right bundle branch block, as well as S waves in lead I, Q waves in lead III, and T waves in lead III.
ECG Findings
ECG findings for PE have poor sensitivity and specificity and are not diagnostic. It is useful to be familiar with some of these findings and to be familiar with the differential diagnosis associated with these fidings. Notably, while ECG findings are not necessarily useful diagnostically, features of RV strain (S1Q3T3, RBBB, TWIs in V1-V4, aVR ST elevation) on ECG are suggestive of a worse prognosis and increased risk of shock and death.
Common ECG findings are listed below:
Sinus tachycardia is the most common finding (in around 27-40% of cases).
The classic S1Q3T3 finding is not sensitive or specific for PE, occurring in only 4% of cases4; rather it is a marker of right heart strain in general.
T waves inversions, particularly in V1-V4 (34% of cases).
New rightward axis deviation is another classic but uncommon finding (11% of cases).
Transthoracic Echocardiogram (TTE) Findings
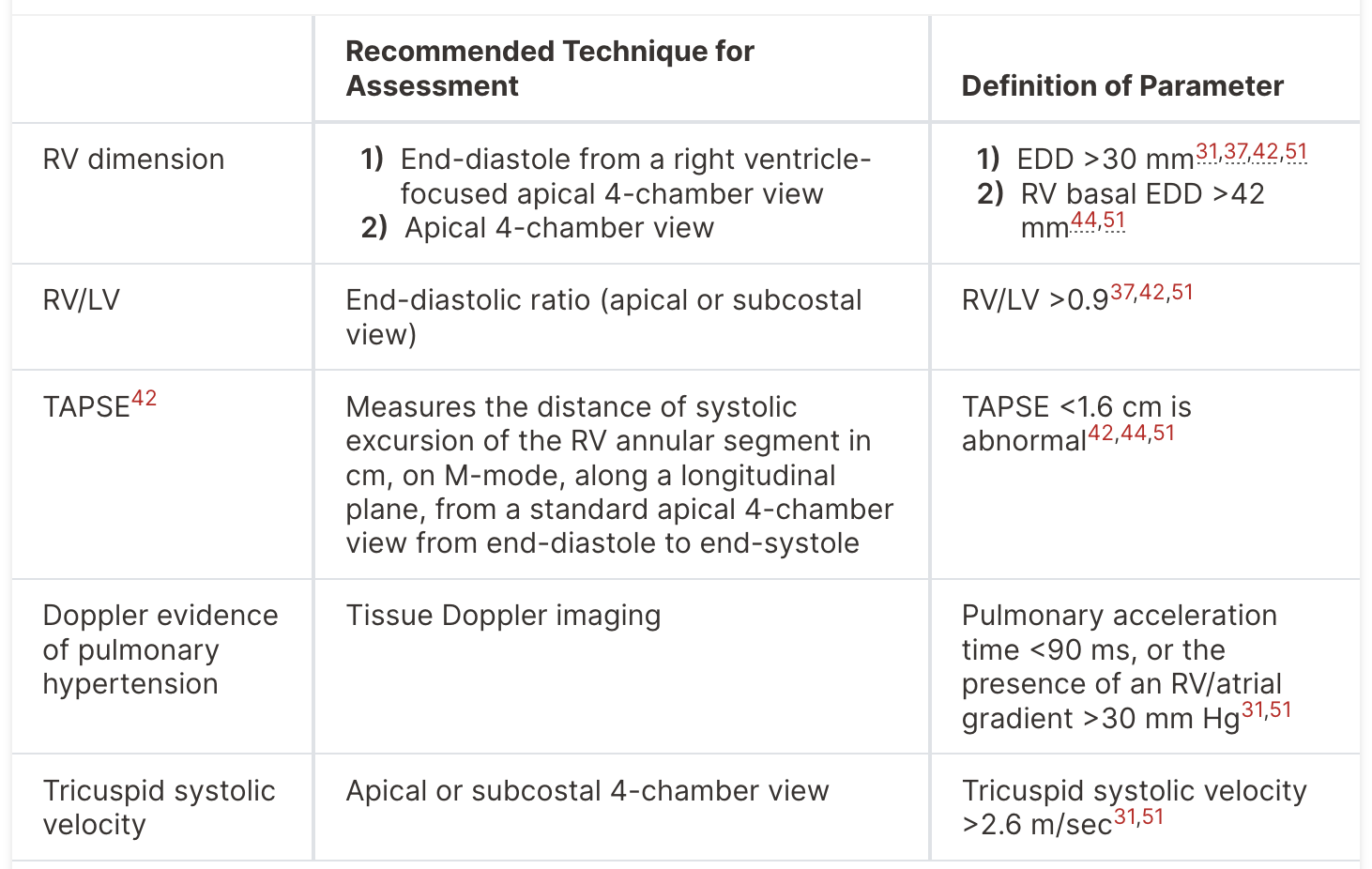
Transthoracic echocardiogram (TTE) and point-of-care ultrasound (POCUS) are useful bedside tools for risk-stratifying intermediate and high-risk pulmonary embolism. However, they are not a reliable diagnostic test, as many patients will have normal TTE findings. TTE / POCUS can be used to (1) detect RV strain and (2) investigate other causes of cardiopulmonary decompensation (e.g. tension pneumothorax, acute MI, pericardial effusion). Identifiing these features via TTE is operator dependent, and should be confirmed with a formal TTE / cardiology review. Once a PE is diagnosed, TTE is an essential part of risk stratification.
Note: elevated pulmonary artery systolic pressure (PASP) and / or RV hypertrophy are signs of chronic pulmonary HTN and potential signs of chronic PE.
This image is a parasternal long axis depicting increased right ventricular (RV) pressure and size. Usually, the left ventricle (LV) should be greater than the RV. However, in this image, the RV is significantly enlarged which is consistent with increased right-sided pressures.
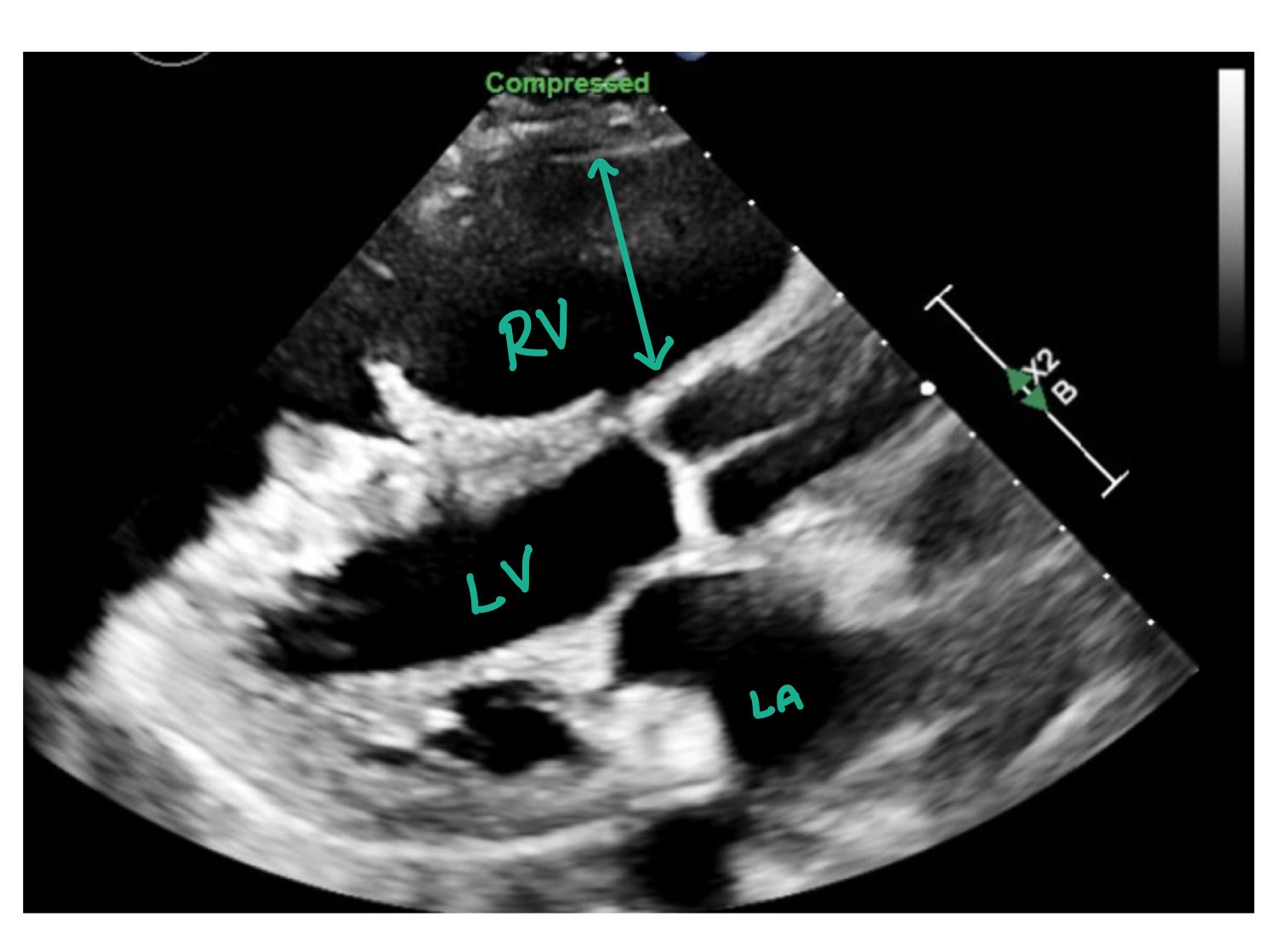
Here is a picture of the parasternal short axis. Again, note how the RV > LV, the D sign shape of the RV during systole with bowing of the intraventricular septum.
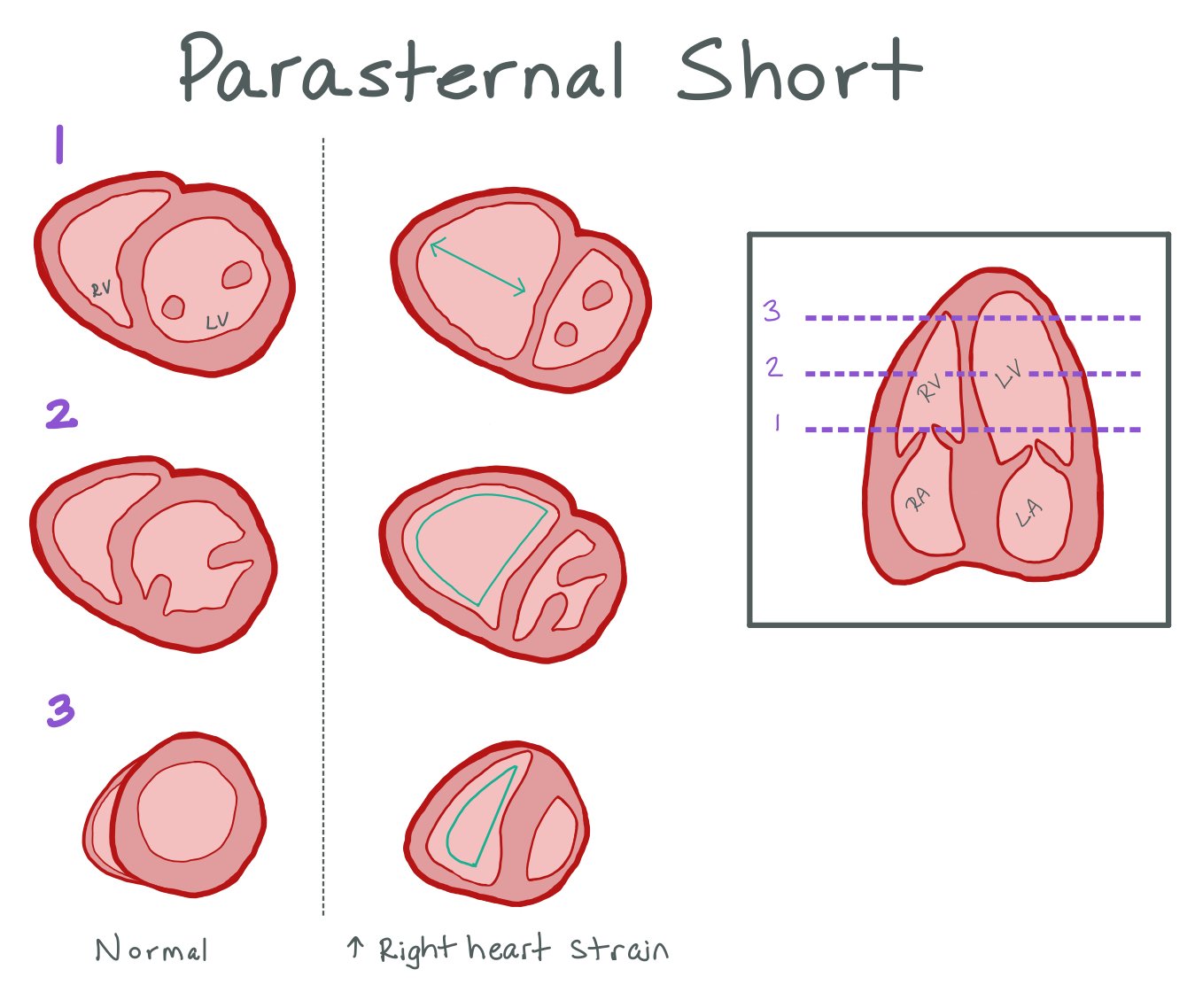
This picture compares the parasternal short views in a normal heart vs a heart with increased RV strain. D Sign occurs when there is RV pressure overload; in the parasternal short view, the LV should normally appear round in diastole and systolic. When there is pressure overload, the LV will appear D shaped throughout the cardiac cycle.
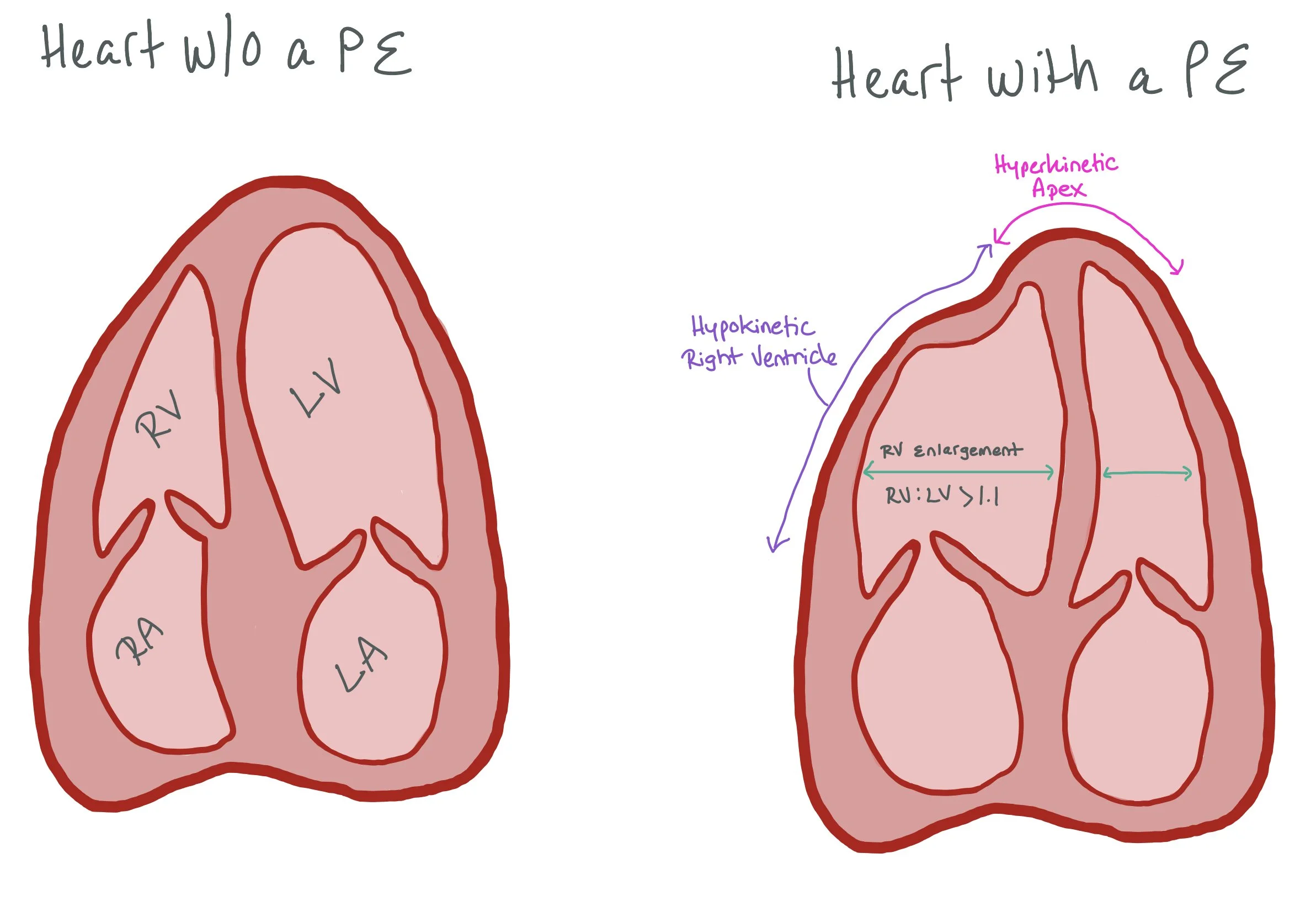
The picture on the left is an apical four chamber view of a normal heart. The picture on the right depicts a heart with a PE. Note the right ventricular (RV) enlargement, hyperkinetic apex, and hypokinetic right ventricle. McConnell’s sign occurs when there is relative hyperkinesis of the RV apex compared to the RV free wall (occurs as the LV apex is hyperkinetic and tethered to the RV apex, causing it to appear hyperkinetic)
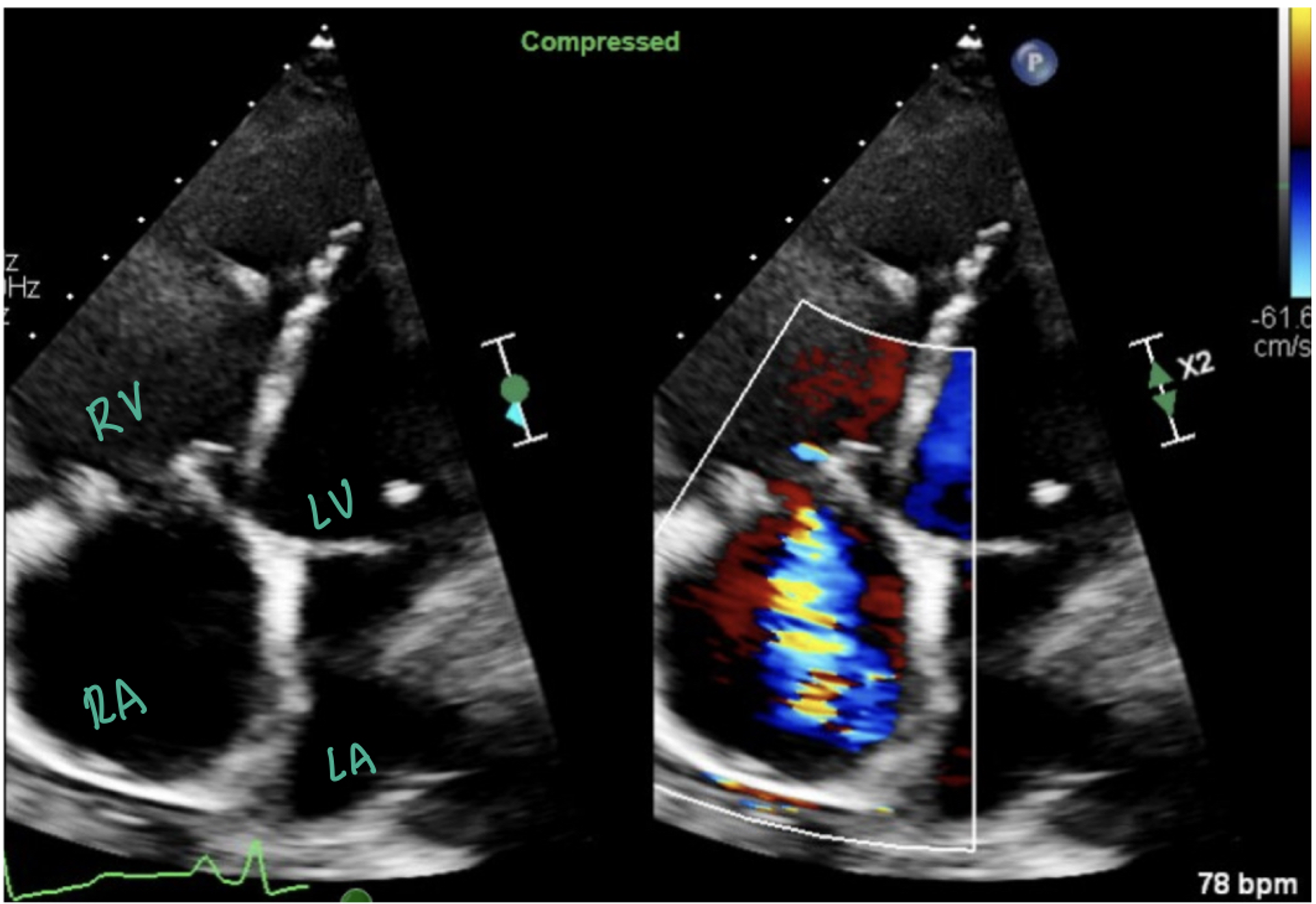
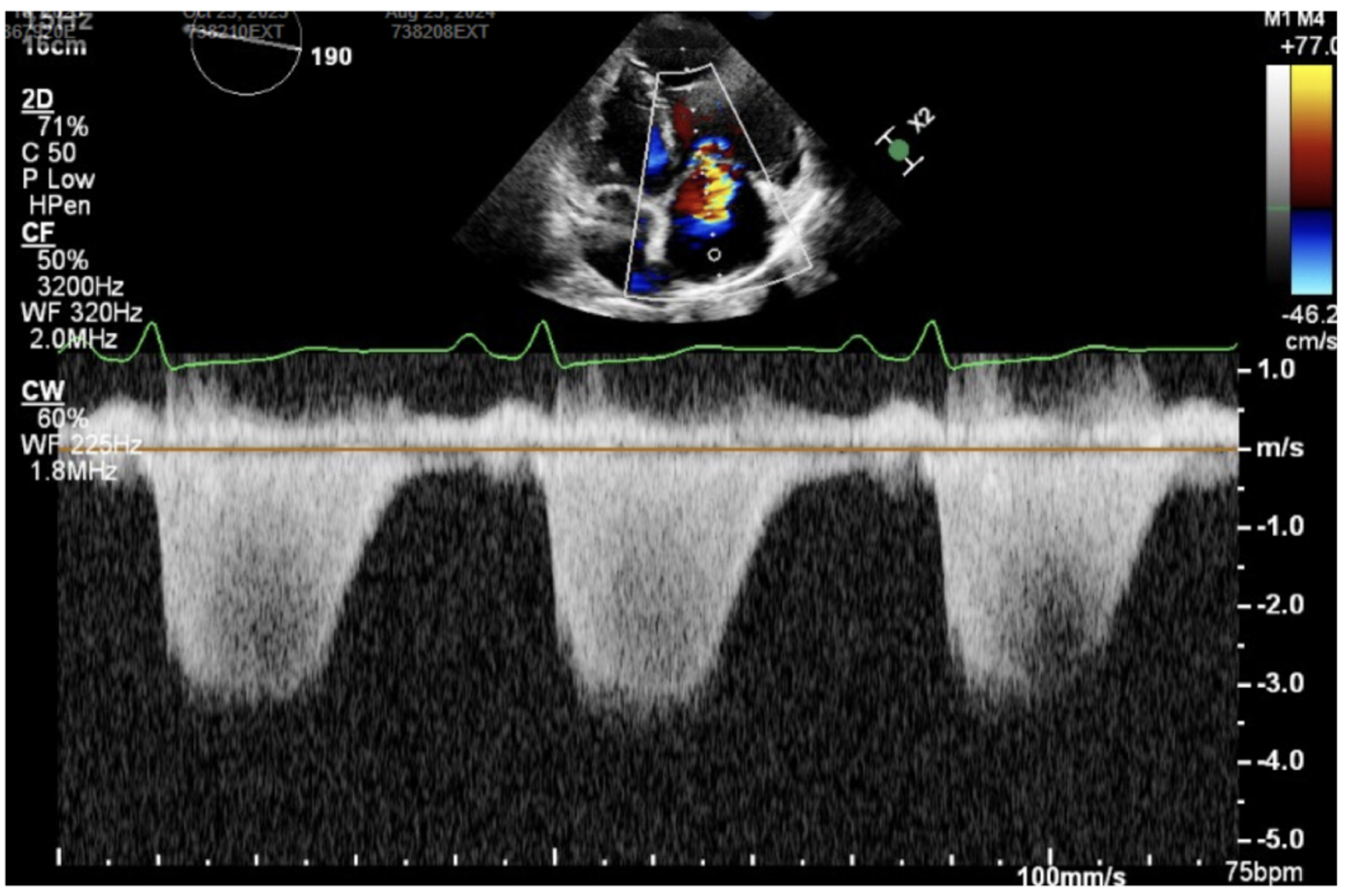
The picture above shows an apical four chamber view with color doppler over the right atrium (RA), showing a tricuspid regurgitation jet into the RA. The picture below shows continuous wave doppler through the tricuspid valve, which is a thick, well-defined curve signifying severe TR.
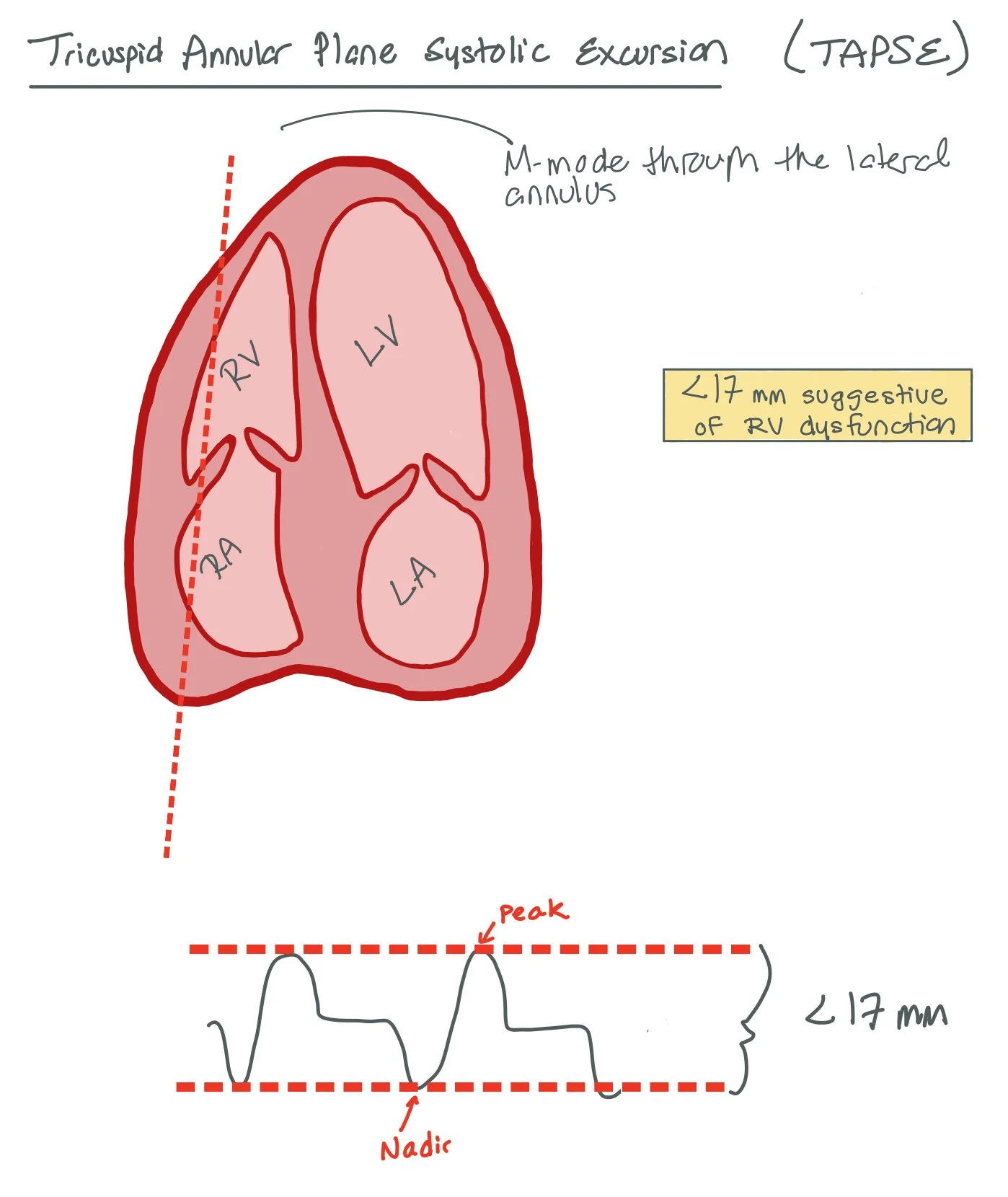
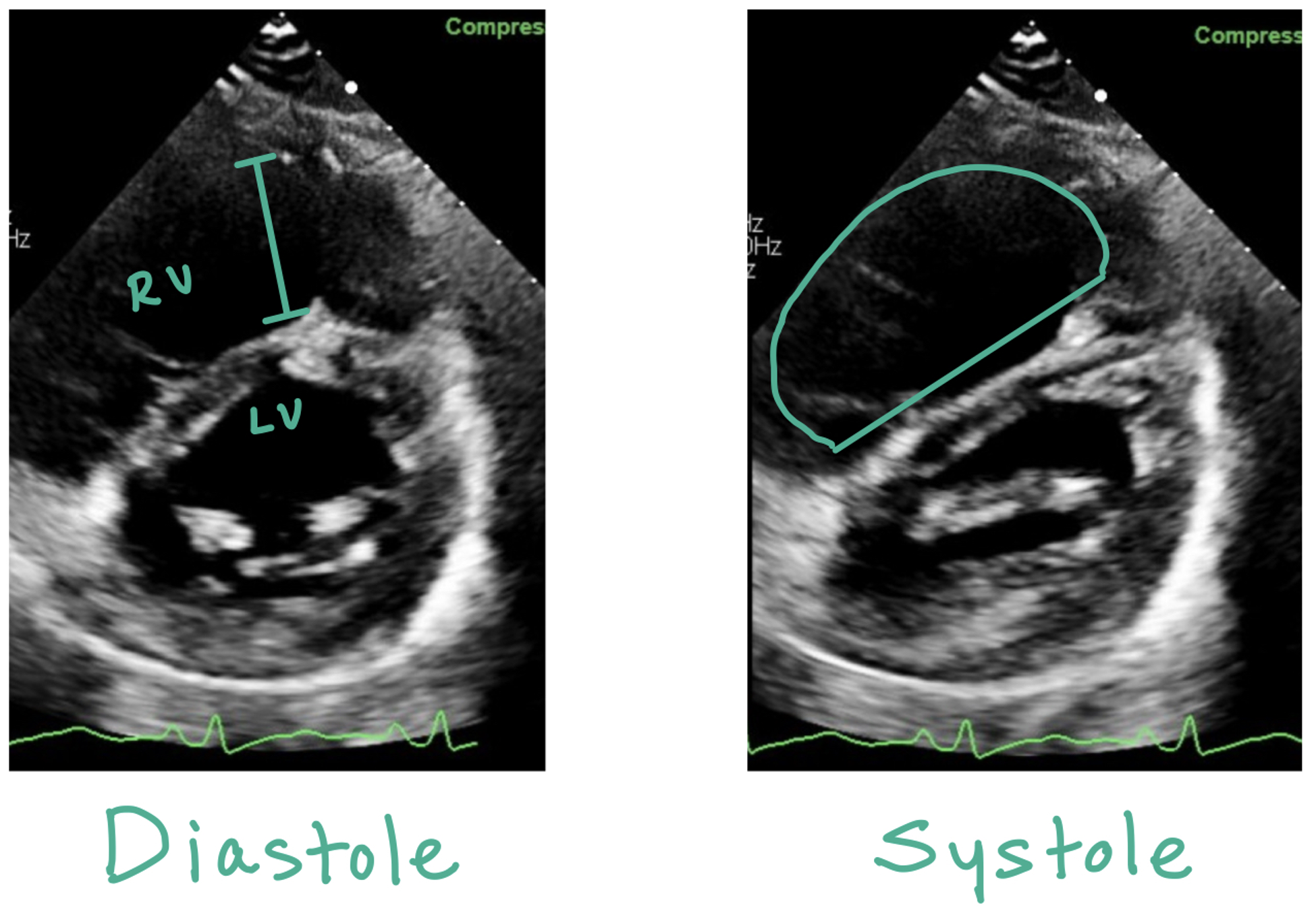
TAPSE (tricuspid annular plane systolic excursion) is assessed with M-mode over the tricuspid annulus in an apical 4-chamber view. This is a surrogate of RV function given the mechanics of RV contraction, which differs from LV contraction. >1.6 cm is a normal finding, while less than this indicates RV dysfunction.
Workup and Classification:
A good history and physical exam are crucial. Patients may experience symptoms of pleuritic chest pain, dyspnea, hemoptysis, and syncope. Physical exam findings include tachypnea, hypotension, hypoxemia, decreased lung sounds, pleural friction rub, JVD, an accentuated pulmonic second heart sound, a parasternal lift, and leg swelling swelling (from DVT). Aspects from the history that make PE more likely are recent surgery, hospitalization, immobility, pregnancy, estrogens, trauma, cancer, inflammatory disorders, and inherited / acquired thrombophilias.
Labs: BNP and troponin can be elevated as pulmonary artery pressures rise increase RV wall tension, leading to RV ischemia and dysfunction that can manifest as RV dilation. Lactic acid may be elevated when a patient has obstructive shock due to PE, leading to end-organ hypoperfusion.
D-dimer: In patients >50 years old, age-adjustment can be calculated by age * 10 to find the upper limit of normal. Elevated D-dimer is >95% sensitive for PE, meaning a negative result is highly unlikely to be a PE. D-dimer is not specific and is often elevated in patients with systemic illness, an issue commonly seen in hospitalized patients.
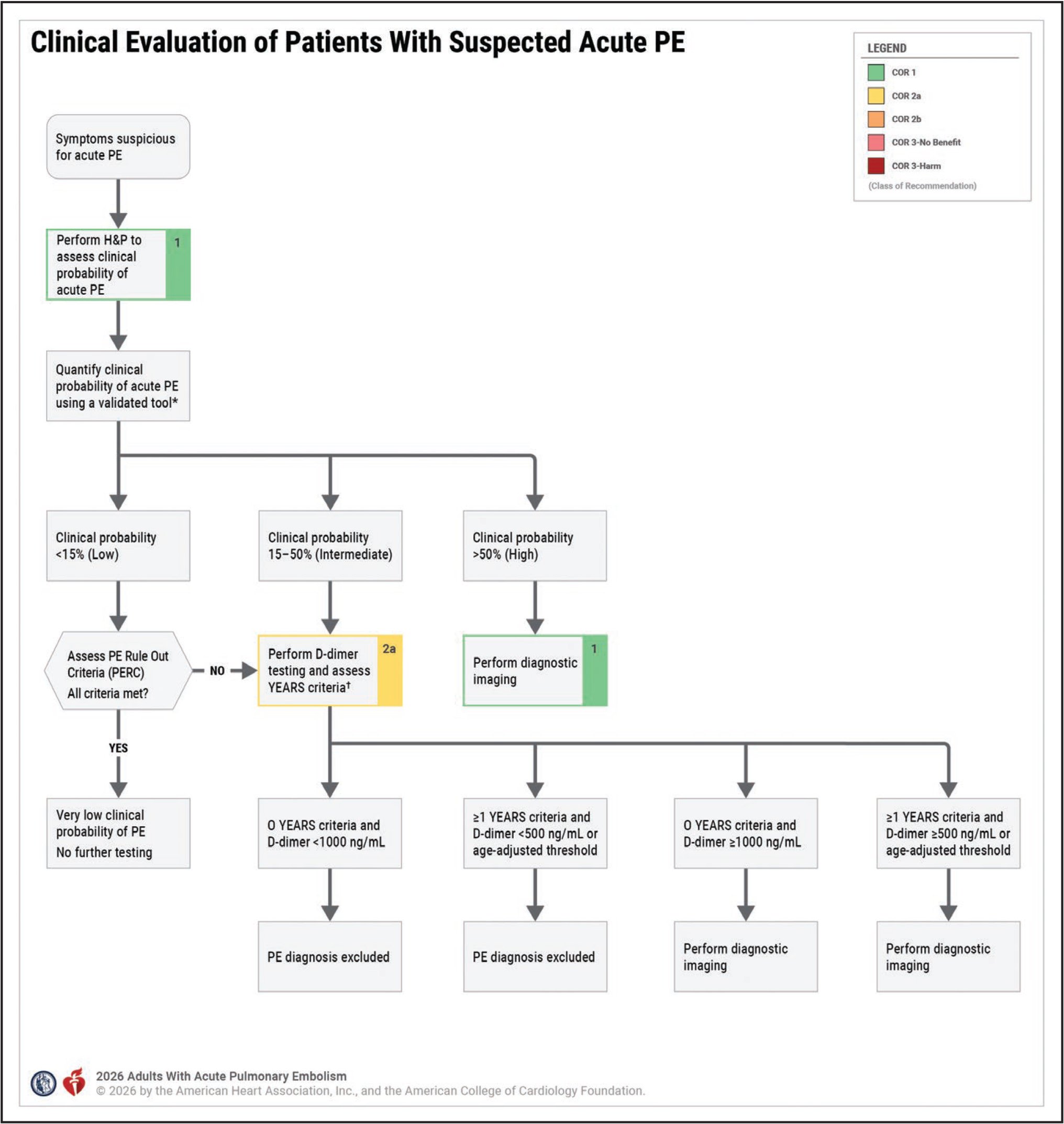
Clinical probability scores: Wells criteria and Revised Geneva score are validated scoring systems used to assess clinical pretest probability. The Geneva score has the advantage of being objective, without the “PE is #1 diagnosis OR equally likely” category found in the Wells criteria. The Pulmonary Embolism Rule-Out Criteria (PERC) is used to rule out PE in low risk patients.
Imaging Options: The first line imaging study is a CT pulmonary angiogram (CTPE/CTPA). Ventilation-perfusion (V/Q) SPECT is a second-line test, often used when contrast exposure is undesirable, such as in patients with significant renal dysfunction, or who have contraindications to radiation such as in pregnancy. The performance of ventilation perfusion SPECT scans for PE diagnosis is not well established2 and the result often ends up being “intermediate risk” for PE, which can complicate decision making.
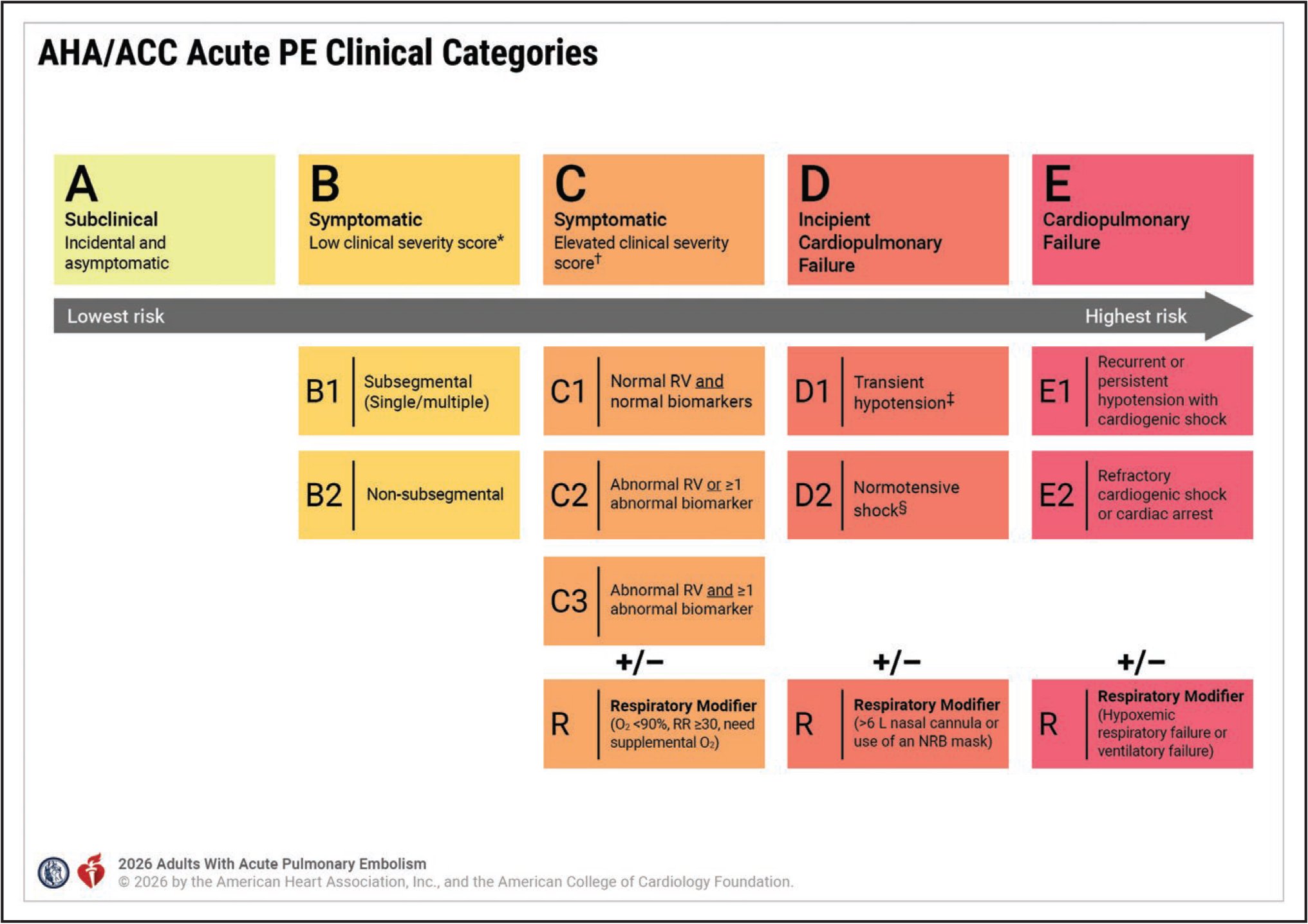
Prior to the new 2026 guidelines, PEs were classified as massive (aka high risk) or submassive (aka intermediate risk) based on size and hemodynamic effects. Now, we will be classifying PEs into 5 categories of A-E and subcategories based on risk to better diagnose and treat acute PE.
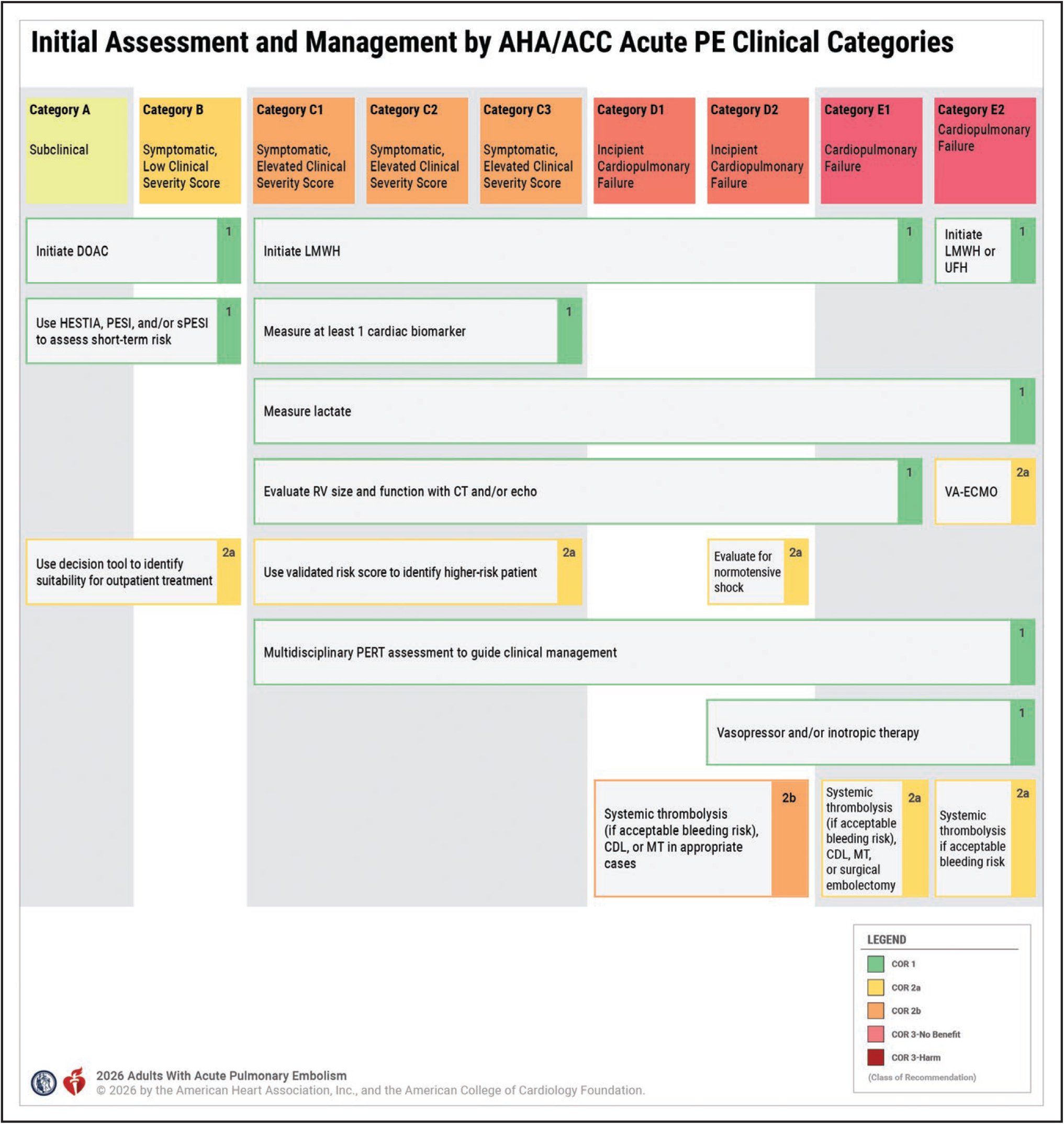
Patients who are Category A and B can just be started on DOAC with no additional labs or imaging. Note: DOACs are now considered superior to VKA. These patients can also be treated outpatient and do not require hospitalization.
Another change to the 2026 guidelines is that patients in Category C-E should be started on LMWH over heparin.
Treatments and Interventions:
Thrombectomy: This method uses a transcatheter device to mechanically remove proximal clots with the main theoretical advantage being avoidance of the significant bleeding risks of thrombolytics. At this time, despite the theoretical benefits of these techniques, supportive date are slim and mostly observational. The FLAME study10 is an observational study suggesting an association with the benefit of mechanical thrombectomy in high-risk PE patients. PEERLESS I was a randomized trial that compared mechanical thrombectomy to catheter-directed thrombolysis, which showed patients with mechanical thrombectomy had lower rates of clinical deterioration and post-procedure ICU time compared with catheter-directed thrombolysis. In contrast, PEERLESS II is an ongoing trial set to end in the summer of 2026 and compares thrombectomy plus anticoagulation to anticoagulation alone.
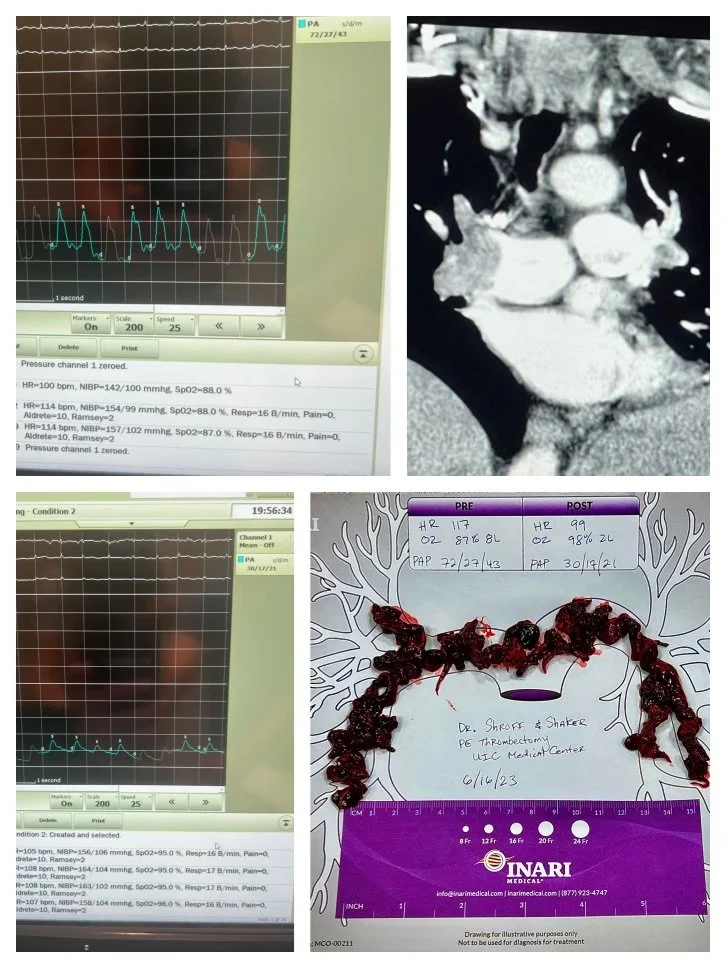
Catheter Directed Thrombolytics allow for reduced TPA dose (12mg or 24mg) which is directly injected into the pulmonary artery. Theoretically, this method may be more effective in clot dissolution and may reduce risk of bleeding; however, in brief, data to support these theoretical benefits is very scant12. It is unclear if there is any benefit to catheter-directed thrombolytics versus an equivalent IV dose.
** Beyond the bleeding risks associated with thrombolytics and systemic anticoagulation for PE, transcatheter interventions carry additional risks for procedure-related bleeding, such as access site hematoma, retroperitoneal bleed, and peripheral or pulmonary vascular injury. Other device-specific complications are varied, and the comparative risk-benefit data is still preliminary.
Surgical Thrombectomy is an open surgical procedure in which a cardiothoracic surgeon removes large clots from the pulmonary arteries to rapidly restore blood flow to the lungs, typically using cardiopulmonary bypass. It is generally reserved for high-risk cases where patients have hemodynamic instability, shock, or cardiac arrest—especially when standard treatments like systemic thrombolysis are contraindicated or have failed. Evidence for surgical thrombectomy in PE comes largely from observational studies, registries, and improvements in surgical technique. Contemporary guidelines from organizations, such as the American Heart Association and the European Society of Cardiology, support its use in carefully selected patients with high-risk PE, particularly when rapid clot removal is necessary and less invasive catheter-based options are unavailable or unsuitable. Overall, while not first-line for most PE cases, surgical thrombectomy remains a critical, potentially life-saving option in severe presentations when other therapies cannot be used or are ineffective.
Thrombolytics: For patients Category C and D, systemic thrombolytics is a 2B recommendations. For patients with Category E, thrombolytics has 2A recommendation.
Anticoagulation:
Low molecular weight heparin is the preferred form of AC in patients with Category C-E.
Patients in Category A and B should be started on DOACs, while all patients should ideally be put on DOACs prior to discharge rather than VKA medications. The main exception to this is patients with anti-phospholipid syndrome (APLS), who should receive wawrfarin, and pregnant patients who should receive low molecular weight heparin.
Duration of Anticoagulation:
For patients with reversible causes of PEs (such as recent surgery, trauma, prolonged travel), it is reasonable to give them anticoagulation for 3-6 months.
For patients without reversible causes (i.e. active cancer, iatrogenic, etc), these patients should be on anticoagulation beyond 6 months. The updated guidelines suggest that these patients continue on a maintenance half dose of medication (i.e. apixaban 2.5mg BID or xarelto 10mg) indefinitely rather than full dose anticoagulation.
If there is concern that a patient developed another clot while on AC, this should be assessed through additional imaging. If they were on half dose AC when this occurred, they should continue back on full dose anticoagulation.
Patients with genetic conditions, such as APLS, should continue on therapeutic warfarin indefinitely.
Which do you pick? It all depends on the stability of the patient and what experts at each institution are comfortable performing. Patients in Categories A and B should receive AC alone, while patients in Categories C-E may require additional therapy, such as catheter directed thrombolytics, thrombectomy, etc.
Testing for Hypercoagulable or Prothomboic States
Patients who have had a PE should have close follow-up within the first week of discharge from the hospital to ensure appropriate medication adherence, detect bleeding, and closely follow symptoms. At every visit within the first year, the patient’s symptoms should be assessed. For those who have unresolved symptoms, we must ensure they do not have Chronic Thromboembolic Pulmonary Disease (CTEPD), a primary lung condition that causes multiple PEs to form (occurs in 2-4% of patients with acute PE).
Each of these patients should have age appropriate cancer screening tests (occurring in 4-10% of patients with acute PE without major reversible risk factors).
For patients without any major reversible factor who develop PE and are < 55 years, screening should be done to assess for genetic and acquired thrombophilia. Prior to ordering these genetic tests, shared decision making should be made. It is important to note that per the new guidelines, thrombophilia testing is not recommended in patients who had a PE associated with a major reversible risk factor, like surgery, trauma, or immobilization. Thrombophilia does not usually increase the risk for recurrence and does not usually influence duration of anticoagulation.
Deficiencies in anticoagulants, such as antithrombin, protein C, and protein S, are linked to higher risk of recurrence and may need longer durations of anticoagulation. These coagulation-based tests should NOT be performed during the acute phase of a PE as it will give you a false result!!
Pulmonary Embolism in Special Populations
Substantial disparities exist in PE diagnosis and access to care.
Access to advanced PE therapies—including catheter-based interventions, ECMO, and Pulmonary Embolism Response Teams (PERTs)—varies widely by institution. Even when these resources are available, PE remains underdiagnosed and undertreated in specific populations, including women, Black patients, individuals with cancer, and those with underlying thrombophilias, reflecting persistent gaps in clinical recognition and representation in clinical trials.
Cancer-associated VTE requires individualized, cancer-specific management.
Venous thromboembolism is the second leading cause of death in patients with malignancy. While most cancer-associated VTE can be treated with direct oral anticoagulants for a minimum of six months, the choice and duration of therapy must be tailored to cancer type, bleeding risk, and ongoing oncologic treatment. Particular caution is required in high-risk malignancies, such as primary or metastatic CNS tumors.
Pregnancy presents distinct diagnostic and therapeutic challenges in PE.
Pregnancy significantly increases VTE risk, yet pregnant patients are largely excluded from randomized trials, resulting in major evidence gaps. PE accounts for 11% of deaths among pregnant patients. Diagnostic evaluation should prioritize radiation minimization, with lower-extremity ultrasound as the initial test when appropriate. Trimester-adjusted D-dimer thresholds and the modified YEARS algorithm can aid risk stratification. Despite frequent imaging, only approximately 2% of studies ultimately confirm PE, underscoring the need for judicious testing.
Family history strongly predicts VTE risk, while thrombophilia testing has limited utility.
A first-degree family history of VTE is a powerful independent risk factor. Standard thrombophilia testing identifies only about half of inherited disorders and should not be performed in the acute setting. When indicated, genetic testing offers greater diagnostic accuracy. Testing for homocysteine levels and MTHFR mutations is not recommended due to a lack of clinical utility.
Race, obesity, and hormone therapy substantially modify VTE risk.
PE incidence is higher in Black populations and lower in Asian populations. Obesity is a major risk factor, with a marked increase in VTE risk at BMI ≥40 kg/m². Oral estrogen therapies—including contraceptive and gender-affirming regimens—are associated with increased VTE risk, whereas transdermal estrogen appears safer. Depot medroxyprogesterone acetate also confers increased risk and should be considered carefully when selecting contraceptive options.
Back to the Case:
What tools can be used to guide the appropriate use of diagnostic imaging when PE is on the differential diagnosis?
A good history and physical exam are crucial. Patients may experience symptoms of pleuritic chest pain, dyspnea, hemoptysis, and syncope. Physical exam findings include tachypnea, hypotension, hypoxemia, decreased lung sounds, pleural friction rub, JVD, an accentuated pulmonic second heart sound, a parasternal lift, and leg swelling swelling (from DVT). Aspects from the history that make PE more likely are recent surgery, hospitalization, immobility, pregnancy, estrogens, trauma, cancer, inflammatory disorders, and inherited / acquired thrombophilias. The modified Geneva Score or Wells Criteria can be used to then further risk stratify patients.
How are patients with PE risk-stratified, and how does this guide further treatment?
In the new guidelines, we categorize patients from A-E with additional subcategories which becomes important for management and treatment. Patients in A and B category can be given a DOAC and sent home, while patients C-E should be started on LMWH and admitted to the hospital. Patients in C-E category should also get additional labs and imaging to help risk stratify the PE.
What is the role of catheter-based interventions for the treatment of PE?
At this time, there is no conclusive data that shows that catheter directed thrombolytics or mechanical thrombectomy is better than systemic anticoagulation. The more hemodynamically unstable the patient is and the higher risk the clot is deemed to be, the more one can argue to perform one of these procedures. In patients who are extremely unstable, ECMO can be an option.
How do we determine anticoagulation duration after a PE?
For patients with reversible causes of PEs (such as recent surgery, trauma, prolonged travel), it is reasonable to give them anticoagulation for 3-6 months. For patients without reversible causes (i.e. active cancer, iatrogenic, etc), these patients should be on anticoagulation beyond 6 months. The updated guidelines suggest that these patients continue on a maintenance half dose of medication (i.e. apixaban 2.5mg BID or xarelto 10mg) indefinitely rather than full dose anticoagulation.
Resident Responsibilities:
Appropriate use of biomarkers and imaging studies, along with vital signs and clinical stability are crucial tools to risk-stratify patients with PE.
When a high or intermediate-high risk PE is identified (aka Category C-E), early involvement of a PERT team / interventional cardiologists is important to determine if a patient may benefit from catheter-based therapies.
Determining anticoagulation duration depends on the risk of recurrence associated with the initial PE and, in many cases, depends on shared decision-making weighing risks of recurrence and bleeding.
The role of thrombophilia testing is overall ill-defined but often does not change management and should not be ordered until 3-6 months AFTER the patient’s initial PE.
Any genetic testing ordered should be done as shared decision making with the patient as it has longterm consequences.
Patients with PE should be followed up 1 week after discharge and then continuously for the next year to ensure appropriate anticoagulation adherence, no bleeding, and no return of PE.
Additional Learning:
2026 ACC / AHA Pulmonary Embolism Guideilnes
Stay tuned for CardioNerds PE Series! Here’s the first episode in the series: https://www.cardionerds.com/443-pulmonary-embolism-the-modern-approach-to-pulmonary-embolism-care-with-dr-kenneth-rosenfield/
How’d we do?
The following individuals contributed to this topic: Mohsin Dahodwala, MD; Catherine Vanchiere, MD; Megan Burke, MD; Pal Shah, MD
Chapter Resources
1. Creager MA, et al. 2026 AHA/ACC guideline for the management of patients with acute pulmonary embolism: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2026;153:e00–e00. doi:10.1161/CIR.0000000000001415
2. Special Populations of PE -- CardioNerds
3. Use of Intravenous Iodinated Contrast Media in Patients with Kidney Disease: Consensus Statements from the American College of Radiology and the National Kidney Foundation Matthew S. Davenport, Mark A. Perazella, Jerry Yee, Jonathan R. Dillman, Derek Fine, Robert J. McDonald, Roger A. Rodby, Carolyn L. Wang, and Jeffrey C. Weinreb Radiology 2020 294:3, 660-668
4. Kahn S, de Wit K. Pulmonary embolism. N Engl J Med. 2022;387(1):45-57. DOI: https://doi.org/10.1056/NEJMcp2116489.
5. Loscalzo, Joseph, and Joseph Loscalzo. “Deep-Venous Thrombosis and Pulmonary Thromboembolism.” Harrison’s Principles of Internal Medicine, Twenty-First Edition (Vol. 1 & Vol. 2), 21st ed., McGraw-Hill Education, 2022, pp. 2091–2101.
6. Mattu, Amal. “Amal Mattu's ECG Case of the Week – December 3, 2018.” ECG Weekly, 3 Dec. 2020, https://ecgweekly.com/weekly-workout/ems-ecgs-s1q3t3-is-it-pathognomonic-for-acute-pulmonary-embolism/.
7. Academic Emergency Medicine 2015; 22: 1127–1137 © 2015 by the Society for Academic Emergency Medicine
8. Connors, J.M. Thrombophilia Testing and Venous Thrombosis. N. Engl. J. Med. 2017, 377, 1177–1187.
9. Khairani, C, Bejjani, A, Piazza, G. et al. Direct Oral Anticoagulants vs Vitamin K Antagonists in Patients With Antiphospholipid Syndromes: Meta-Analysis of Randomized Trials. J Am Coll Cardiol. 2023 Jan, 81 (1) 16–30. https://doi.org/10.1016/j.jacc.2022.10.008
10. Baumgartner C, Klok FA, Carrier M, et alClinical Surveillance vs. Anticoagulation For low-risk patiEnts with isolated SubSegmental Pulmonary Embolism: protocol for a multicentre randomised placebo-controlled non-inferiority trial (SAFE-SSPE)BMJ Open 2020;10:e040151. doi: 10.1136/bmjopen-2020-040151\
11. Wang, Chen et al. “Efficacy and safety of low dose recombinant tissue-type plasminogen activator for the treatment of acute pulmonary thromboembolism: a randomized, multicenter, controlled trial.” Chest vol. 137,2 (2010): 254-62. doi:10.1378/chest.09-0765
12. Silver MJ, Gibson CM, Giri J, et al. Outcomes in High-Risk Pulmonary Embolism Patients Undergoing FlowTriever Mechanical Thrombectomy or Other Contemporary Therapies: Results From the FLAME Study. Circulation: Cardiovascular Interventions. 2023;16(10). doi:10.1161/circinterventions.123.013406
13. Giri J, Sista AK, Weinberg I, et al. Interventional Therapies for Acute Pulmonary Embolism: Current Status and Principles for the Development of Novel Evidence: A Scientific Statement From the American Heart Association. Circulation. 2019;140(20). doi:10.1161/cir.0000000000000707
14. Meyer G. Fibrinolysis for Patients with Intermediate-Risk Pulmonary Embolism. The New England Journal of Medicine. Published April 10, 2014. https://www.nejm.org/doi/full/10.1056/NEJMoa1302097
15. Rivera-Lebron B, McDaniel M, Ahrar K, Alrifai A, Dudzinski DM, Fanola C, Blais D, Janicke D, Melamed R, Mohrien K, Rozycki E, Ross CB, Klein AJ, Rali P, Teman NR, Yarboro L, Ichinose E, Sharma AM, Bartos JA, Elder M, Keeling B, Palevsky H, Naydenov S, Sen P, Amoroso N, Rodriguez-Lopez JM, Davis GA, Rosovsky R, Rosenfield K, Kabrhel C, Horowitz J, Giri JS, Tapson V, Channick R; PERT Consortium. Diagnosis, Treatment and Follow Up of Acute Pulmonary Embolism: Consensus Practice from the PERT Consortium. Clin Appl Thromb Hemost. 2019 Jan-Dec;25:1076029619853037. doi: 10.1177/1076029619853037. PMID: 31185730; PMCID: PMC6714903.
16. Smith B MD. Right Heart Strain - Core Ultrasound. Core Ultrasound - free and subscription ultrasound education for medical professionals. Published June 28, 2021. Accessed April 9, 2024. https://coreultrasound.com/right-heart-strain/
17. Avila J MD. Right Heart Function - Core Ultrasound. Core Ultrasound - free and subscription ultrasound education for medical professionals. Published February 14, 2020. Accessed April 9, 2024. https://coreultrasound.com/right-heart-function/
18. Avila J MD. 60/60 Sign for Acute Pulmonary Embolism - Core Ultrasound. Core Ultrasound - free and subscription ultrasound education for medical professionals. Published February 14, 2020. Accessed April 9, 2024. https://coreultrasound.com/60-60-sign-for-acute-pulmonary-embolism/